Abstract
Introduction:
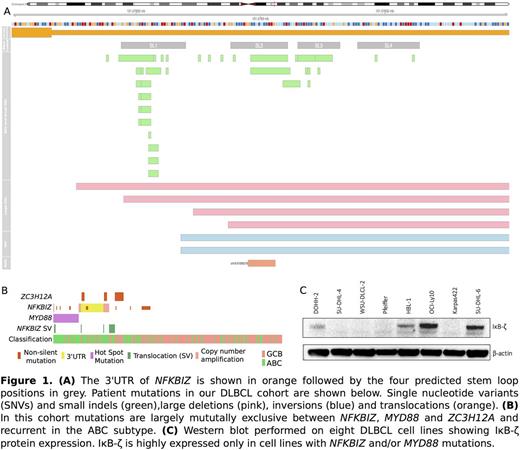
Diffuse large B-cell lymphoma (DLBCL) represents the most frequent non-Hodgkin lymphoma, accounting for over 30% of cases. The two main subtypes of DLBCL, germinal center B-cell-like (GCB) and activated B-cell-like (ABC), have distinct clinical outcomes, unique gene expression profiles and a growing list of defining genetic features. Recurrent somatic mutations affecting genes such as MYD88, CD79A/B and TNFAIP3 have been shown to constitutively activate NF-κB signaling, either directly or indirectly, in ABC DLBCL. Amplification of the NFKBIZ locus has been observed in ~10% of ABC DLBCL cases however, no other mutations affecting this locus have been previously described. Although some alterations leading to NF-κB activation have been identified and characterized, there remain ABC cases with NF-κB activation and no clear genetic explanation. Through a meta-analysis of published and in-house exome and genome sequence data for recurrent mutations affecting non-coding regions, we found NFKBIZ had the strongest enrichment for mutations within the 3' untranslated region (UTR). The majority of these mutations affect a highly conserved region of the mRNA that is predicted to form multiple stem-loops or more complex secondary structures (Figure 1A). The endoribonuclease Regnase-1, encoded by the gene ZC3H12A, targets mRNAs with conserved secondary structures in the 3'UTR such as those predicted to form in NFKBIZ . Mutations in the NFKBIZ 3'UTR potentially abrogate this interaction with Regnase-1.
Aims and Methods:
Based on the observed mutation pattern, we hypothesized that NFKBIZ 3'UTR mutations are more significantly enriched in ABC DLBCL and lead to increased activation of the NF-κB signaling pathway. We aimed to (1) determine the frequencies of mutations in these two genes in DLBCL, specifically within each sub-type in our cohort and (2) investigate the effects of NFKBIZ 3'UTR mutations on mRNA and protein expression in activating the NF-κB signaling pathway in B-cells. We analyzed RNA-seq data and performed targeted sequencing on these lines and 334 primary DLBCL tumors and 8 DLBCL cell lines. We assessed mRNA abundance and the allelic abundance of mutant and wild-type NFKBIZ using RNA-seq reads and a custom "wild-type negative" or "drop-off" droplet digital PCR (ddPCR) assay. The selected cell lines represent those naturally NFKBIZ mutant (amplification or containing a 3'UTR deletion), MYD88 mutant, or wild-type for both. These lines were used to investigate NFKBIZ mRNA and IκB-ζ protein expression.
Results:
Targeted sequencing and copy number analysis of 316 cases with available RNA-seq data uncovered 66 DLBCLs (21% of cases) containing any NFKBIZ mutation, 34 (10.8%) of which were 3'UTR mutations, and 19 DLBCLs with a mutation in ZC3H12A (6% of cases) . A substantial fraction of the NFKBIZ mutations were confirmed somatic by sequencing matched constitutional DNA. NFKBIZ 3'UTR mutations were mutually exclusive with MYD88 mutations and largely segregated from those affecting ZC3H12A (Figure 1B). In aggregate, mutations in either of these novel genes are present in 22.6% of all DLBCLs and 30.8% of ABC cases. The enrichment for these mutations in ABC cases was highly significant (OR 4.1, 95% CI 2.04-8.48). Results from our ddPCR experiments revealed allelic imbalance of NFKBIZ mRNA in mutant cell lines, specifically a relative increase of the mutant transcript. Patient RNA samples harboring NFKBIZ mutations (small deletions and SNVs) showed similar results. Western blot experiments confirmed increased levels of IκB-ζ protein consistent with increased mRNA in all NFKBIZ and MYD88 mutant cell lines (Figure 1C).
Conclusion:
Our current work has shown promising results indicating that NFKBIZ may be an underappreciated driver gene in ABC DLBCL. We have demonstrated that mutations affecting the 3'UTR of the NFKBIZ gene are significantly enriched in the ABC sub-type. These mutations cause deregulation of the mRNA and lead to over-expression of the IκB-ζ protein which activates the NF-κB signaling pathway. Uncovering new ways in which the NF-κB pathway can be dysregulated in ABC DLBCL will allow for better treatments to be developed and more accurate stratification of patients into the clinical trials that will be the most beneficial to their specific set of mutations.
Connors: Bristol-Myers Squibb: Research Funding; Genentech: Research Funding; Merck: Research Funding; Lilly: Research Funding; Cephalon: Research Funding; F Hoffmann-La Roche: Research Funding; Janssen: Research Funding; Bayer Healthcare: Research Funding; Seattle Genetics: Research Funding; Takeda: Research Funding; NanoString Technologies, Amgen, Bayer, BMS, Cephalon, Roche, Genentech, Janssen, Lilly, Merck, Seattle Genetics, Takeda,: Research Funding; Amgen: Research Funding; NanoString Technologies: Research Funding. Scott: Janssen: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; BCCA: Patents & Royalties: Patent describing molecular subtyping of DLBCL licensed to NanoString Technologies. Patent describing measurement of the proliferation signature in MCL.. Steidl: Affimed Therapeutics: Consultancy; Juno Therapeutics: Consultancy. Morin: Epizyme, Inc: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal